Photoacids have been known for more than 70 years. Theodor Förster has been the first to correctly describe the observations of absorption and fluorescence spectra of photoacids, and connect positions of the electronic transitions giving rise to optical absorption bands to the increased acidity properties of photoacids in the electronic excited state. Many research activities have been pursued in the following decades, but apart from quantum chemical calculations of photoacid molecules of medium size, focussing on the intramolecular electronic charge distribution changes of the proton donating moieties of photoacids, microscopic insight have remained limited. Some of these studies have indicated – in line with previous suggestions based on physical organic principles – that the effects of electronic excitation are much more pronounced on the conjugate photobase side of the Förster cycle (Fig. 1).
An ultrafast X-ray glance into photoacid electronic structure
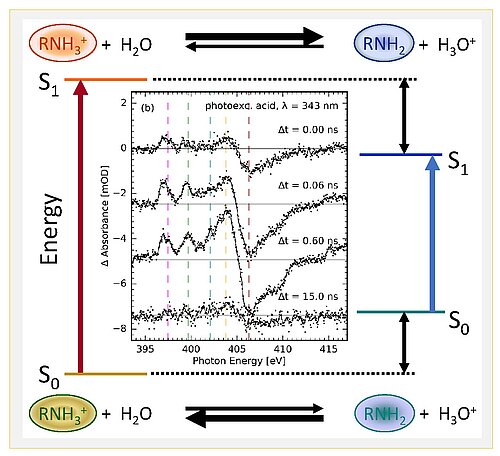
Fig. 1: Förster cycle of an amine photoacid, showing electronic ground states S0 and the first excited states S1 of the acidic (left) and basic (right) species. There are the four stages of photoacid behaviour in aqueous solution, as shown by the cartoons. In the centre transient soft-X-ray spectra are shown, measured on 8-aminopyrene-1,3,6-trisulfonate (APTS).
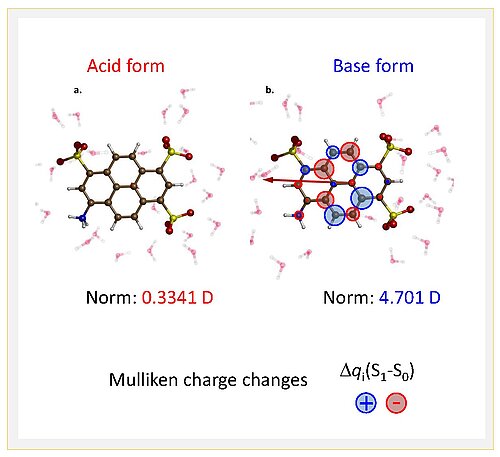
Scientists from the Max Born Institute in Berlin, Stockholm University, the University of Hamburg, Helmholtz-Zentrum Berlin, Ben-Gurion University of the Negev in Beersheva and Uppsala University, have now successfully pursued a novel combined experimental and theoretical approach to study the electronic charge distributions of photoacids along the four stages of photoacids provide direct microscopic insight into the electronic structural changes of the proton donating amine group of an aminopyrene derivative in aqueous solution. The K-edge X-ray absorption spectra of nitrogen atoms in the molecular structure were measured at the synchrotron BESSY II in transmission mode to locally probe electronic structure on ultrafast time scales. Together with quantum chemical calculations, such results provide a consistent picture of photoacid behaviour (Fig. 2): electronic charge distributions of the proton donating group are only minor on the photoacid side, but substantial on the conjugate photobase side. Yet the overall dipole moment change of the whole molecule is as important as the local charge distribution changes, hence solvation dynamics by the solvent water is the second important factor governing photoacidity.